Introduction
In ceramic-to-metal assemblies, engineers often focus heavily on brazing strength, metallization quality, or hermeticity testing. However, one issue that repeatedly appears in real production is surprisingly basic: the nickel plating suddenly peels off.
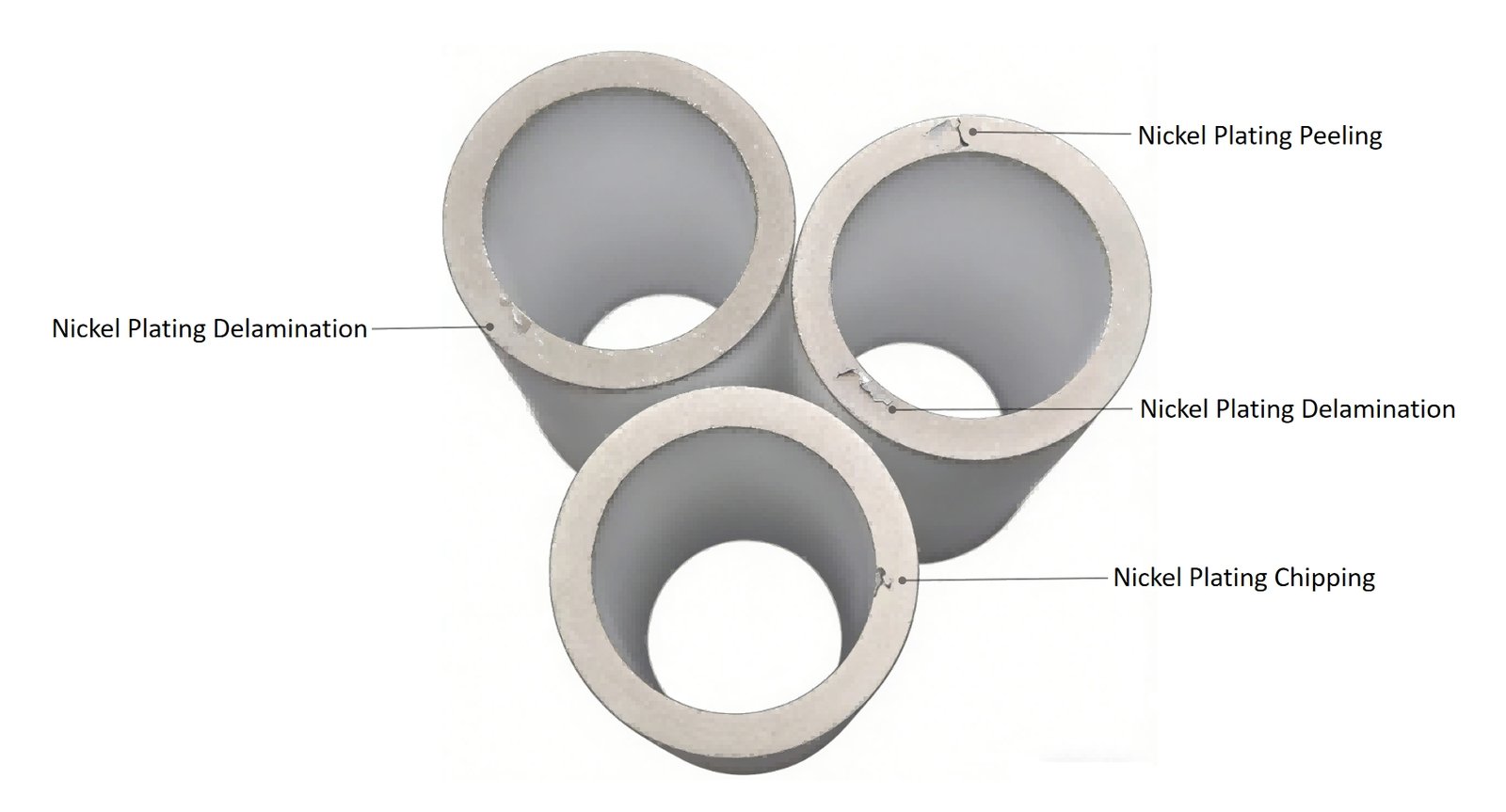
Sometimes the problem shows up immediately after plating. More frustrating cases appear later — during brazing, leak testing, thermal cycling, or even after the customer has already assembled the component into a device.
We have seen cases where the nickel layer looked perfectly normal under visual inspection, passed tape testing, and even survived shipping, only to blister and separate after the first heating cycle. In practice, this usually means the real defect was already present much earlier in the process.
A common misunderstanding is that nickel plating delamination is purely an electroplating defect. In reality, the plating bath is often only the “last step that exposes the problem.” The root cause may originate from cleaning, metallization sintering, storage conditions, or thermal expansion mismatch.
This article summarizes several of the most common causes we encounter in production of metallized ceramics, along with some practical observations from actual manufacturing experience.

Four Common Causes of Nickel Plating Delamination
Inadequate Surface Preparation Before Plating
This is probably the most underestimated cause.
In theory, the metallized surface entering the plating line should be chemically clean and fully activated. In reality, contamination happens more often than many people expect. Fingerprints, polishing residue, residual oil from fixtures, or even dust absorbed during storage can interfere with nickel deposition.
What makes this tricky is that the parts may initially look acceptable. The plating appears smooth and continuous, so the defect remains hidden until later thermal exposure.
Once the assembly goes through brazing or elevated-temperature service, trapped contaminants can volatilize or expand. The resulting local stress often creates blistering or edge lifting between the nickel layer and the metallization layer.
Activation is another overlooked detail. Alumina ceramics are more sensitive to incomplete activation because residual glass phase on the surface can reduce active bonding sites. If the activation step is too mild — or too short — nickel deposition may begin unevenly, creating weak interfacial regions from the start.
Problems in the Metallization Layer Itself
In many failure cases, the nickel layer is not the primary problem at all.
If the metallization layer has poor density, excessive porosity, or weak bonding to the ceramic body, the nickel simply adheres to an already unstable surface.
Typical issues include:
- Uneven metallization thickness
- Excessive glass phase bleeding onto the surface
- Weak Mo-Mn sintering structure
- Oxidized metallization surfaces after storage
- Microcracks generated during metallization sintering
When the metallization layer contains excessive porosity, the nickel cannot form an effective mechanical interlock. The deposited metal may bridge over pores instead of anchoring into the structure.
Storage conditions also matter more than many factories admit. Metallized ceramic parts stored too long in humid air can gradually form oxide films or absorb contamination. Even when oxidation is not visually obvious, it can significantly reduce plating adhesion.
Another issue occasionally seen in production is oversintering. Excessively aggressive sintering parameters may partially vitrify the surface, reducing roughness and active metallic exposure. Ironically, a smoother-looking metallization surface can sometimes produce weaker nickel adhesion.
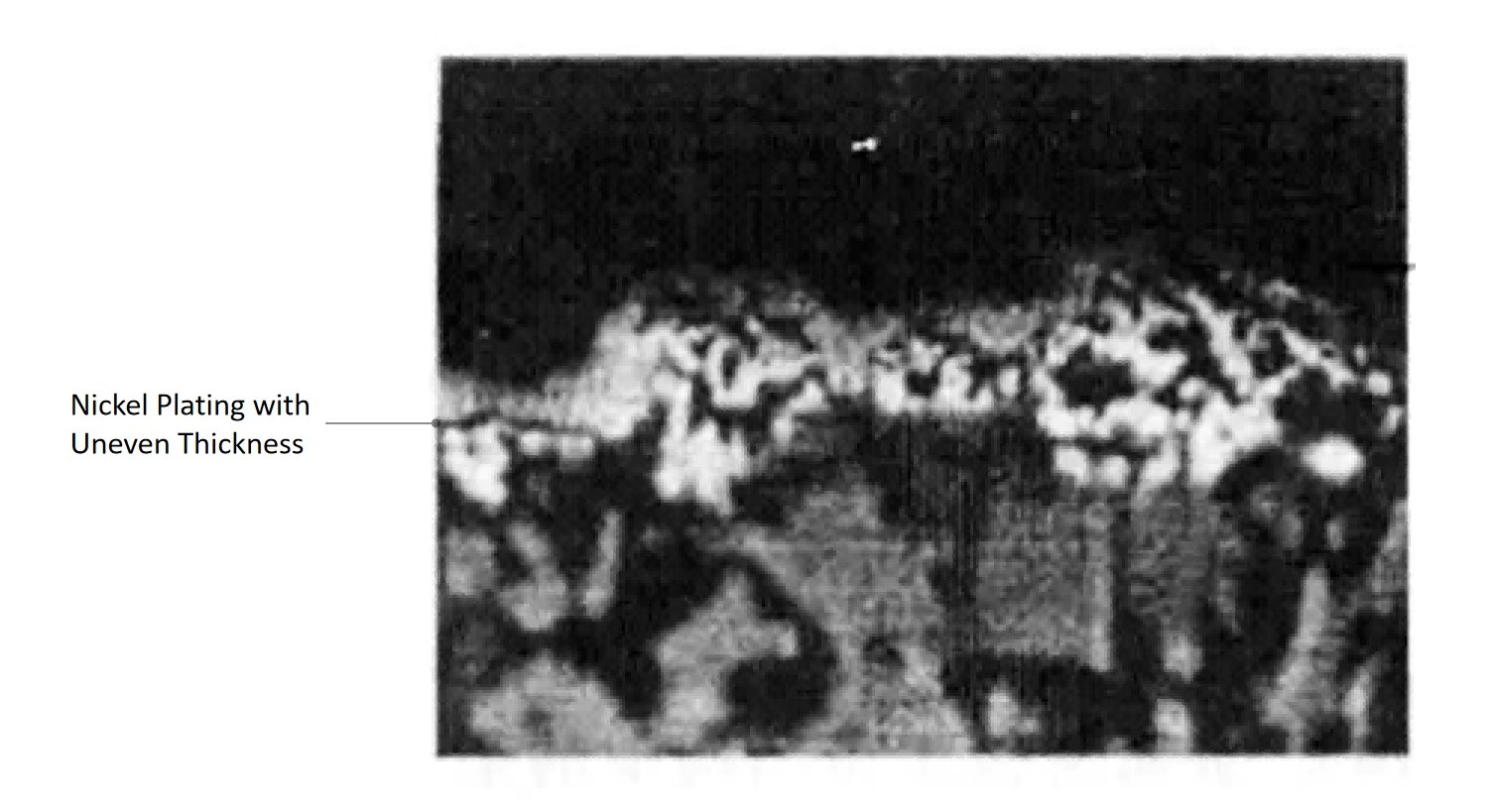
Instability in the Electroplating Process
Even with excellent pretreatment and metallization quality, unstable plating parameters can still generate internal stress strong enough to cause delamination later.
Nickel plating is highly sensitive to process control. Small parameter drift over time may not immediately create visible defects, but it can gradually increase residual stress inside the deposit.
Some common examples are shown below:
| Process Issue | What Usually Happens |
| Excessive current density | Nickel deposits too quickly, forming coarse columnar grains and higher internal stress. |
| Large pH fluctuations | Deposit becomes porous, brittle, or chemically unstable. |
| Poor bath contamination control | Embedded impurities weaken the continuity of the nickel layer. |
| Temperature instability | Crystal growth becomes inconsistent, increasing stress concentration. |
| Aging plating solution | Additive imbalance affects deposit ductility and adhesion. |
Thermal Stress After Plating
Even when plating adhesion is initially good, thermal expansion mismatch can still create problems later.
Nickel, Mo-Mn metallization, ceramic bodies, and brazing fillers all expand differently during heating. When thermal stress exceeds the local bonding strength, interfacial separation begins.
This becomes especially critical in:
- Thick nickel plating
- Large-area metallized surfaces
- Rapid heating or cooling cycles
- High-temperature brazing applications
- Repeated thermal cycling environments
The mismatch is particularly noticeable between nickel and alumina ceramics because their CTE differ substantially.
For example, if a thick nickel layer is deposited mainly to improve solderability, the additional metal thickness may unintentionally increase stress accumulation during thermal cycling.

Practical Solutions for Improving Adhesion
Improving Pre-plating Cleaning and Activation
Good adhesion starts long before electroplating.
In actual production, a combination of chemical cleaning and ultrasonic cleaning is usually more reliable than relying on chemical degreasing alone. Ultrasonic agitation helps remove contaminants trapped inside microscopic surface pores and metallization texture.
Another practical improvement is minimizing the delay between cleaning and plating. Even short exposure to humid workshop air can affect activated surfaces.
For difficult materials or older metallized surfaces, mild micro-etching before plating can significantly improve activation consistency.
Optimizing Metallization and Sintering
A stable metallization layer is the real foundation of reliable nickel plating.
Several process details are especially important:
- Proper Mo-to-glass ratio in the metallization paste
- Controlled sintering atmosphere
- Stable wet hydrogen dew point
- Uniform metallization thickness
- Controlled surface roughness after sintering
The goal is not simply achieving conductivity. The metallization layer must also provide sufficient mechanical anchoring sites for the plated nickel.
In practice, excessively smooth metallization surfaces are not always desirable.
Storage management after metallization sintering is equally important. Many manufacturers underestimate how quickly oxidation or contamination can occur after sintering.
Stabilizing the Electroplating Process
Consistent process control is more important than aggressively pursuing faster deposition speed.
The following ranges are commonly used in nickel plating control:
| Parameter | Typical Control Range | Risk if Uncontrolled |
| pH | 4.1–4.6 | Brittle deposits, pinholes, poor adhesion |
| Temperature | 55–60℃ | Grain coarsening and stress increase |
| Current density | 2–5 A/dm² | Columnar grain growth and cracking |
| Agitation stability | Moderate and uniform | Uneven deposition thickness |
In real production environments, plating bath maintenance discipline often matters more than theoretical parameter settings written in process documentation.
Controlling Post-Plating Handling and Heating
The process does not end once plating is completed.
Hydrogen embrittlement relief treatment is often necessary after nickel plating, especially for components that will later experience brazing or thermal cycling.
Gradual heating is also critical. Fast temperature ramps can create thermal shock at the interface long before the entire assembly reaches equilibrium temperature.
For sensitive hermetic components, vacuum storage or inert-gas packaging is sometimes used to minimize oxidation before brazing assembly.
Conclusion
Nickel plating delamination on metallized ceramics is rarely caused by a single isolated factor.
In most real-world cases, the failure develops from a chain of smaller process deviations: slight contamination during cleaning, marginal metallization density, unstable plating stress, combined with thermal expansion mismatch during later assembly.
That is why some parts pass incoming inspection but still fail after brazing or thermal cycling months later.
From our experience, solving delamination problems usually requires looking at the entire manufacturing sequence rather than only adjusting plating parameters.
A plating layer that survives long-term thermal and mechanical stress depends on the combined stability of metallization, surface preparation, electroplating, and thermal process control.
Frequently Asked Questions
Q1: Is nickel plating delamination usually caused by plating defects or metallization defects?
A1: Either is possible, but metallization problems are often overlooked. If the metallization layer itself has weak bonding, oxidation, or excessive porosity, even a well-controlled nickel plating process may still fail later.
Q2: Does aluminum nitride require a different nickel plating process than alumina?
A2: Yes. Aluminum nitride generally requires more careful surface activation because its surface chemistry differs from alumina. In practice, plating parameters and pretreatment methods are often adjusted separately for AlN ceramics.
Q3: Can pinholes in the nickel layer eventually lead to delamination?
A3: They can. Pinholes may allow moisture, corrosive media, or thermal stress concentration to reach the underlying metallization layer. Over time, this can weaken local adhesion and initiate peeling.
Q4: Does nickel plating delamination affect hermeticity?
A4: In many cases, yes. Once the plating separates, underlying pores or cracks in the metallization layer may become exposed, potentially creating leakage paths in hermetic assemblies.
Q5: Can delaminated nickel plating be repaired?
A5: Sometimes. If the problem is limited to the nickel layer itself, stripping and replating may be possible. However, if the metallization layer is already damaged or partially detached from the ceramic, rework is usually unreliable.




